 For DNA/RNA, that might be a complementary nucleic acid sequence that is labeled in some fashion (radioactivity or dye). Last, a visualizing agent specific for the molecule of interest in the mixture is added to the membrane. The membrane may be treated to covalently link the bands to the surface of the blot. The transfer can be accomplished by diffusion or by using an electrical current to move the molecules from the gel onto the membrane.  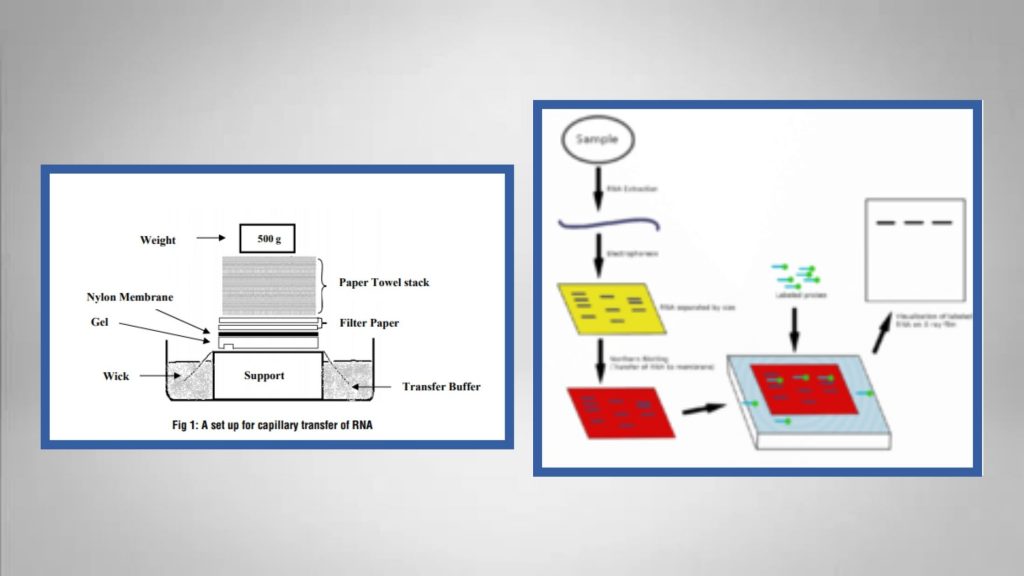
This “blot", as it is called, has an imprint of the bands of nucleic acid or protein that were in the gel (see figure at left). Second, after the gel run is complete, the proteins or nucleic acids in the gel are transferred out of the gel onto a membrane/paper that physically binds to the molecules. The mixture could be DNA (Southern Blot), RNA ( Nothern Blot), or protein ( Western Blot) and the gel could be agarose (for DNA/RNA) or polyacrylamide (for protein). First, the mixture of molecules is separated by gel electrophoresis. In Generi Biotech, we have sound expertise in Southern blotting supported by years of experience and we will take care of the whole process, starting from DNA extraction to probe design.\)īlotting provides a means of identifying specific molecules out of a mixture. In addition to that, a typical assessment of result can be performed by comparison to the customer´s reference sample (as stated above). Testing results from the EOPC and WCB are compared to those of the MCB to allow the detection of any changes that may be indicative of cell line instability. Testing of the working cell banks (WCB) may also be performed at a manufacturer’s discretion, especially if there were any changes in the production process. The minimal characterization required is to assess the Master Cell Bank (MCB) and the End of Production Cell Bank (EOPC). Southern blots also confirm target identity as matching the hybridization probe to digested DNA sample compared to a molecular weight marker and/or digested reference sample. Southern blot analysis is usually a part of a cell line stability testing package, which is a key component in characterizing production cell banks and is critical in maintaining quality assurance of biologicals/ modern therapeutics derived from bacterial, yeast and mammalian cell cultures.īasically, ICH Guidelines require Southern blots (among other methods) to confirm that the structure of the genomic DNA of a cell bank is stable and unchanged and that no clonal rearrangement has occurred. The next step involves transfer and UV cross-linking of the DNA fragments to a membrane and hybridization with a specific nucleic acid probe, which will identify DNA target regions of interest. Then, a certain amount of DNA is digested by a specific restriction endonuclease and cut DNA samples are separated by agarose gel electrophoresis. 
In this method, DNA is first extracted from the sample and quantified by UV spectrometry. 
Southern blot analysis is one of the oldest methods that is used to analyze DNA. GLP Pre-clinical safety and clinical trial testingĪssay Development, Validation and Transfer Master, Working and End-of-production cell bank testing Retrovirus assay - reverse transcriptase enzyme activity (qPBRT) assay Viral seed qPCR adventitious agents testing ATMPs testing (Cell therapy / Gene therapy / Tissue-engineered therapy testing)Ĭell line authentication - DNA genotyping.Custom assay design, validation and transfer 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
